Why Hard Water Is A Huge PROBLEM and What To Do About It?
Is It Safe To Drink Hard Water?
Is hard water safe to drink? Technically, yes.
Consuming small amounts of magnesium and calcium should not cause you any harm. Especially if the rest of the water is clean and free of any harmful contaminants.
The real issue of “hardness” is the effect and nuisance it causes for your home appliances and plumbing.
This is through things like mineral buildup and the potential for clogging or blocking up pipes.
Disadvantages
![]() The biggest issue with hardness is by far the limescale buildup in your pipes and home appliances.
The biggest issue with hardness is by far the limescale buildup in your pipes and home appliances.
![]() You may find that you use more soap because it takes longer to lather, which can be both time consuming and expensive in the long term.
You may find that you use more soap because it takes longer to lather, which can be both time consuming and expensive in the long term.
![]() The limescale it leaves can make your appliances and machines work less efficiently.
The limescale it leaves can make your appliances and machines work less efficiently.
Advantages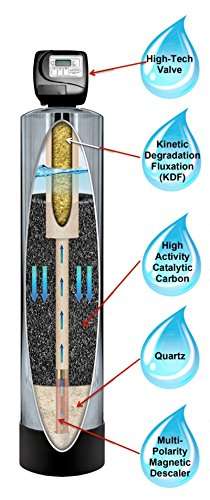
![]() It contains dissolved calcium, it may (at least in theory) be a source of calcium. However, the available calcium in “hard” water is probably not enough to compete with calcium rich foods like leafy vegetables.
It contains dissolved calcium, it may (at least in theory) be a source of calcium. However, the available calcium in “hard” water is probably not enough to compete with calcium rich foods like leafy vegetables.
![]() Acquired taste. Yes this might sound weird but some people actually like the taste of hard water.
Acquired taste. Yes this might sound weird but some people actually like the taste of hard water.
![]() Minimal health risks, unlike soft water. Especially water that has been softened with salt).
Minimal health risks, unlike soft water. Especially water that has been softened with salt).
![]() There are also unconfirmed reports of fewer heart attacks in locations with lots of hardness. It is not clear whether this is as a result of casualty or is simply correlation.
There are also unconfirmed reports of fewer heart attacks in locations with lots of hardness. It is not clear whether this is as a result of casualty or is simply correlation.
What Is Hard Water?
It is water that has high levels of dissolved mineral deposits like magnesium and calcium.
The most visible effect of this is white stains (also known as limescale) around your sink, faucet, bathtub and kitchen appliances like your kettle.
How Is Hard Water Formed?
 It’s formed when magnesium and calcium ions are dissolved in high quantities.
It’s formed when magnesium and calcium ions are dissolved in high quantities.
If the hardness is temporary then this will be due to dissolved calcium hydrogencarbonate and this can easily be removed by boiling it.
However, permanent hardness is due to dissolved calcium sulfate that you can’t simply remove by boiling.
Why Is Hard Water Such A Big Problem?
Hard water is what causes the white limescale stains around your sink, faucet, kettle and bathtub. Do you know that around 90% of the US suffer from it according to The U.S. Geological Survey?
 The situation is almost as bad in other parts of the world like Europe.
The situation is almost as bad in other parts of the world like Europe.
For instance, around 13 million homes in the UK suffer from heavy limescale because of hardness.
All of this is because of the type of water that is supplied to the home. And a lot of this is down to the water available in your geological location.
For instance, living near certain types of rock will determine the kind of water you get.
“Hardness” is not necessarily how water appears in its natural form. For instance, do you know that rainwater is soft in its natural form?
You only get hardness when the water comes into contact with other elements like the ground, chalk, limestone and rock – that its state can change.
Temporary Hard Water?
If your water only contains calcium hydrogencarbonate (instead of calcium sulfate) then it is considered to be “temporary hard”.
“Temporary” because you can easily remove calcium hydrogencarbonate by boiling it unlike other forms of hardness.
Examples Of Hard Water
 High levels of “hardness” means that it is high in both calcium and magnesium.
High levels of “hardness” means that it is high in both calcium and magnesium.
Rain is naturally soft but becomes “hard” when it comes into contact with things like rock, chalk and limestone.
Here are some examples of hard water:
- Water that does not easily lather when you use soap.
- Water that leaves limescale residue in your kettle and pipes (this can cause blockages over time).
- Another example is the type of water that contains ferrous irons. This can result in some reddish brown stains on your clothes after laundry.
Hard Water Areas UK
Unfortunately, the UK is heavily affected with hardness due to its geographic location. About 13 million homes in the UK live in areas with hardness.
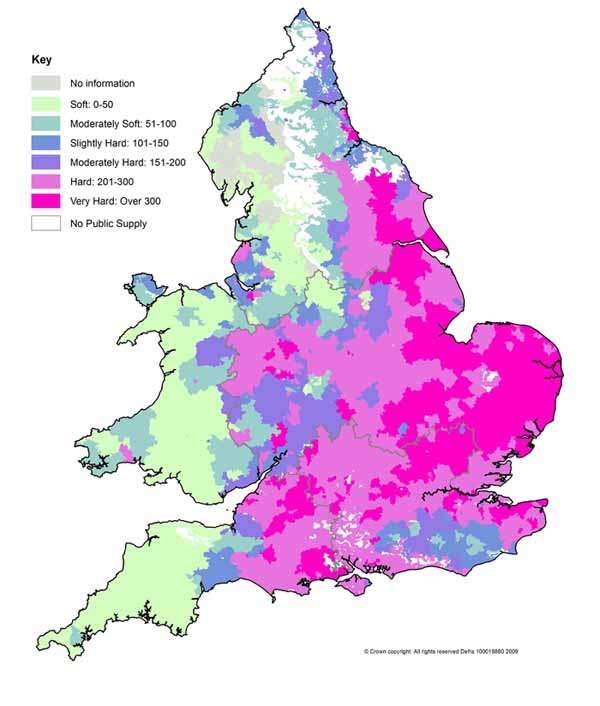 You can check areas suffering from hardness on the UK map and see if your neighbourhood is one of the affected areas.
You can check areas suffering from hardness on the UK map and see if your neighbourhood is one of the affected areas.
 Another way to check hardness is to get a good quality water hardness testing kit.
Another way to check hardness is to get a good quality water hardness testing kit.
It is possible to get a cheap kit but they might not work as well to get you an accurate reading.
Also, remember that you measure hardness by looking at the number of mineral deposits by ppm (parts per million).
Hardness in water is anything more than 180ppm, while anything less than 60ppm can be considered to be soft water.
What Is Soft Water?
This is literally the opposite of hard water in that it doesn’t contain high levels of calcium and magnesium.
In some ways, soft water is actually more natural in the sense that things like rain is naturally soft. Despite this, you shouldn’t drink it if it has been softened with sodium salt. Read why this is the case here.
How To Get Rid of Hard Water?
 While the water from your tap in a western country is usually safe to drink, the hardness can wreck havoc on your pipes, plumbing and appliances.
While the water from your tap in a western country is usually safe to drink, the hardness can wreck havoc on your pipes, plumbing and appliances.
So here is how to get rid of it: soften your water.
The easiest way to do this is to  use a water softener machine.
use a water softener machine.
One of the best softener machines is the Aquasana Whole House Rhino. I like this machine because it not only softens but it filters too.
It softens without using salt or sodium. This is ideal for people who want to consume less sodium in their diet.
If you have very high levels of hard water then consider a salt based softener like this combination machine.
 |  |
| Treats hard water UP TO 25 ppm (low to medium low hard water). | Treats hard water OVER 25 ppm (medium to extremely hard water). |
| Does NOT require electricity. | Requires electricity if using the electronic head monitor. |
| Control Your System With An App:? NO | Control Your System With An App:? YES |
| - Powerful salt-FREE water softener. - 1,000,000 one million gallon capacity. - Aquasana Rhino exceeds NSF standards. - Treats hard water WITHOUT salt. - Removes chlorine, bacteria, iron, sulfide, manganese, mercury, lead etc. - Includes pre-filter, copper-zinc and carbon filtration stages with standard fittings. - 90-Day Pure Satisfaction Guarantee. - Limited 10 year warranty.  | - Powerful salt-BASED water softener. - 1,000,000 one million gallon capacity. - Also includes a multi-stage filter. - Easy installation (inside or outside) - Sufficient for 1-3 bathrooms (options for 4-6 and 7+ bathrooms also available). - 6 month money back guarantee. - Limited lifetime warranty. |
| $$$ Ships to US and CA. | $$$ Ships to US, CA and Puerto Rico. |
| Sale prices automatically applied by clicking this link. | |
| If there is *no* sale try coupon code: AQGEEK50 | Coupon Code: GEEK5 |
 |  |







need Something new to sell my house quickly. Will the cheaper water conditioner work for all water.
Hi Judy
My best advice is to get a water test kit to understand exactly how hard your water is and if there is anything else in your water that you need to concered about.
This is one of the cheapest standalone softeners. However, many people find that a better option is to get an all-in-one softener and filter so that it treats all your water in one go.
It might seem more expensive upfront but you will still need to get an additional filter because standalone softeners don’t produce drinkable water.
Stay hydrated,
Luke
Really informative article.. I’ve learned some new info from it. I would say that using electronic water conditioner is one of the best method to soften the hard water and the reason is that it consumes no salt and requires no maintenance as well. The power of removing scales from water is also Excellent.
That’s why people are going for water conditioners these days. The cost is comparatively low as well if compared with any water softener.
Hi Mrs Sarah
Many thanks for offering your opinion on this topic. I agree that some people prefer electronic descalers over regular water softeners because there so many advantages.
Best wishes,
Luke